Presented by: Paris Tranos, PhD, ICOphth, FRCS
Consultant in Vitreoretinal Surgery & Uveitis
Edited by: Penelope de Politis, MD
A 46- year old man presented with progressive bilateral visual loss for one year.
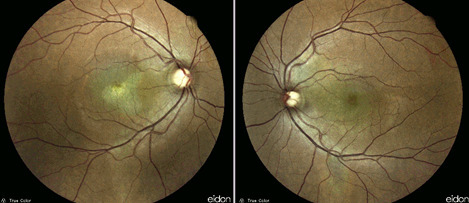
Figure 1
Case History
A 46 year-old caucasian man presented with progressive bilateral visual loss for one year, with partial spontaneous improvement on the left but none on the right. Past medical and family history were unremarkable and he was not on any medication. On examination, best-corrected visual acuity (BCVA) was reduced to counting fingers on the right and 5/10 on the left. Intraocular pressure was within normal limits and there was no sign of inflammation in the anterior or posterior segments. Fundoscopy revealed atrophic lesions and evidence of shallow subretinal fluid at the right and left maculas, respectively (Figure 1).
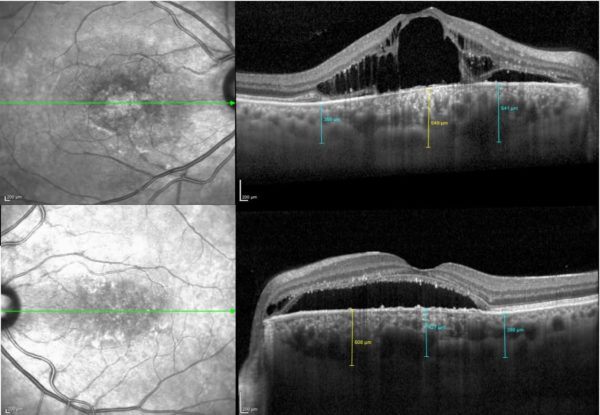
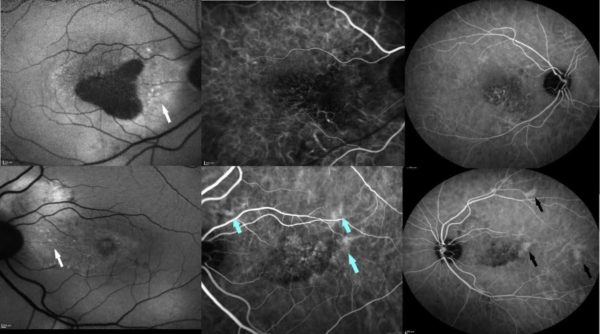
Further investigation with multimodal imaging — autofluorescence (AF), fundus fluorescein angiography (FFA), optical coherence tomography (OCT) and indocyanine green (ICG) angiography — was carried on (Figures 2 and 3).
Figure 2: Spectralis® OCT (Heidelberg Engineering) showing intraretinal and subretinal fluid on the right macular region and subretinal fluid at the left macula, extending up to the optic disc. A small pocket of intraretinal fluid is noticeable adjacent to the left papilla. The choroid is significantly thickened in both eyes, with more pronounced thickening nasal to the macula on the left.
Figure 3: Fundus autofluorescence is decreased at the right macula due to retinal pigment epithelium (RPE) atrophy. Stipple hyper-autofluorescence is evident at the peripapillary area in both eyes (white arrows). Indocyanine green angiography shows pachyvessels (blue arrows) in early phase and multifocal hyperpermeability (black arrows) in midphase.
Additional History
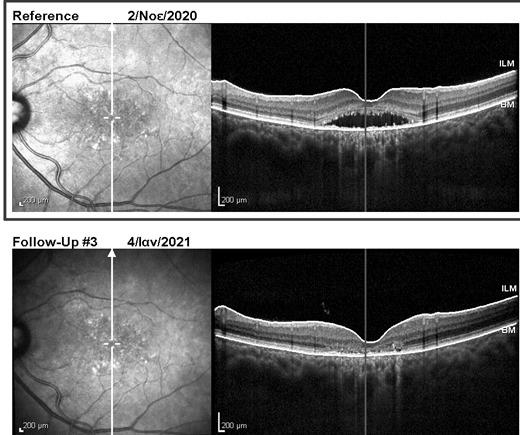
Due to the severe atrophic changes at the macula, the right eye had poor visual prognosis and was managed with observation only. Half-dose photodynamic therapy was applied to the left eye, with complete resolution of subretinal and intraretinal fluid at both macular and peripapillary areas 2 months later (Figure 4). Nevertheless, BCVA change was minimal, improving only to 6/10 after fluid absorption on the left.
Figure 4
Differential Diagnosis
- Peripapillary pachychoroid syndrome
- Other causes of macular edema (diabetic retinopathy, retinal vein occlusion, etc.)
- Pachychoroid neovasculopathy
- Ocular tumors (circumscribed choroidal hemangioma, choroidal hamartoma)
- Cavitary optic disc anomalies (optic pit, advanced glaucoma, coloboma)
- Tractional maculopathy
The peripapillary disposition, associated with nasal choroidal thickening and peripapillary IRF point to the diagnosis of Peripapillary Pachychoroid Syndrome.
Discussion and Literature
A structurally and functionally intact choroid tissue is vitally important for retinal function. While the central retinal artery supplies the internal 2 thirds of the retina, the choroidal vein network is responsible for the remaining external third. Abnormal choroidal blood flow leads to photoreceptor dysfunction and death.1 A wide range of choroidal diseases manifest with increased choroidal thickness, thus being referred to as Pachychoroid Spectrum Disorders, a relatively new concept in ophthalmology (2013), still evolving both in concept and in terminology.2
Pachychoroid Spectrum Disorders (PSD) classification has been constantly revised and is presently divided in six disorders, sometimes coexisting in the same eye:3 Pachychoroid Pigment Epitheliopathy, Central Serous Chorioretinopathy (CSC), Pachychoroid Neovasculopathy, Polypoidal Choroidal Vasculopathy, Focal Choroidal Excavation and Peripapillary Pachychoroid Syndrome (PPS) — these two being the latest additions. They all share 3 basic characteristics: increased choroidal thickening, pathologically dilated veins in Haller’s (outer choroid) layer — pachyvessels — and attenuation of the choriocapillaris and Sattler’s (middle choroid) layer.4
Peripapillary Pachychoroid Syndrome was introduced as a variant of CSC, but is now considered a separate syndrome, due to its particular features, notably the tendency to surround the optic nerve. Also, patients with PPS are mostly men but typically older than those with CSC. Cardinal findings are peripapillary intraretinal fluid (IRF) and/or subretinal fluid (SRF), extending to the nasal macula, with thickening of the nasal macular choroid — as opposed to temporal in CSC. Besides, IRF is more prevalent than SRF and appears earlier in the course of the disease than in CSC. Additional characteristics of PPS include choroidal folds, peripapillary RPE mottling and pachyvessels on ICG. A relatively short axial length and hyperopia are common associations with this disorder.
Peripapillary Pachychoroid Syndrome may go unnoticed until there is macular involvement, thus manifesting as marked reduction in visual acuity. The disease is bilateral but involvement may be asymmetric. Diagnosis poses a challenge in identifying PPS among other forms of PDS, as they are believed to represent different manifestations of a common pathogenic process, with overlapping features and possible progression from one to another.5 Multimodal imaging is the recommended approach for distinguishing this new clinical entity, also from disorders such as posterior uveitis and neuro-ophthalmologic conditions sharing similar features, namely disk leakage and edema, both of which may ensue in complicated PPS.6
Management of PPS may be conservative (observation) or include anti-vascular endothelial growth factor (anti-VEGF) injections and/or photodynamic therapy, the latter being suggested as the most effective treatment modality.7 Despite encouraging anatomical results, visual improvement is usually minimal to moderate. Advanced cases may be entirely refractory to treatment.8
Keep in mind
- PPS is a distinct entity and one of the pachychoroid syndromes
- It is important to exclude the presence of choroidal neovascular membrane, ocular tumors and optic disc cavitary lesions prior to establishing the diagnosis of PPS
- Management is similar to that for CSC, PDT being the most effective treatment modality
References
- Akkaya S (2018). Spectrum of pachychoroid diseases. International ophthalmology, 38(5), 2239–2246. https://doi.org/10.1007/s10792-017-0666-4
- Moraru A D, Costin D, Moraru R L, Costuleanu M & Brănișteanu D C (2020). Current diagnosis and management strategies in pachychoroid spectrum of diseases (Review). Experimental and therapeutic medicine, 20(4), 3528–3535. https://doi.org/10.3892/etm.2020.9094
- Manayath G J, Verghese S, Ranjan R& Narendran V (2020). Simultaneous multiple pachychoroid spectrum entities coexisting in the same eye. Indian journal of ophthalmology, 68(10), 2234–2235. https://doi.org/10.4103/ijo.IJO_931_20
- Castro-Navarro V, Behar-Cohen F, Chang W, Joussen A M, Lai T, Navarro R, Pearce I, Yanagi Y & Okada A A (2020). Pachychoroid: current concepts on clinical features and pathogenesis. Graefe’s archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie, 10.1007/s00417-020-04940-0. https://doi.org/10.1007/s00417-020-04940-0
- Cheung C, Lee W K, Koizumi H, Dansingani K, Lai T & Freund K B (2019). Pachychoroid disease. Eye (London, England), 33(1), 14–33. https://doi.org/10.1038/s41433-018-0158-4
- Phasukkijwatana N, Freund K B, Dolz-Marco R, Al-Sheikh M, Keane P A, Egan C A, Randhawa S, Stewart J M, Liu Q, Hunyor A P et al. (2018). PERIPAPILLARY PACHYCHOROID SYNDROME. Retina (Philadelphia, Pa.), 38(9), 1652–1667. https://doi.org/10.1097/IAE.0000000000001907
- Xu D, Garg E, Lee K, Sakurada Y, Amphornphruet A, Phasukkijwatana N, Liakopoulos S, Pautler S E, Kreiger A E, Yzer S et al. (2020). Long-term visual and anatomic outcomes of patients with peripapillary pachychoroid syndrome. The British journal of ophthalmology, bjophthalmol-2019-315550. Advance online publication. https://doi.org/10.1136/bjophthalmol-2019-315550
- Alonso-Martín B, de-Lucas-Viejo B, Gimeno-Carrero M, Ferro-Osuna M & Sambricio J (2020). Diagnosis by multimodal imaging in peripapillary pachychoroid syndrome: A case report. Diagnóstico mediante imagen multimodal en síndrome paquicoroideo peripapilar: a propósito de un caso. Archivos de la Sociedad Española de Oftalmología, 95(5), 248–253. https://doi.org/10.1016/j.oftal.2020.01.013