Presented by: Evangelos Lokovitis MD, FEBOphth
Edited by: Penelope Burle de Politis, MD
A 50-year-old man was referred for management of a slowly progressive proptosis on the right.
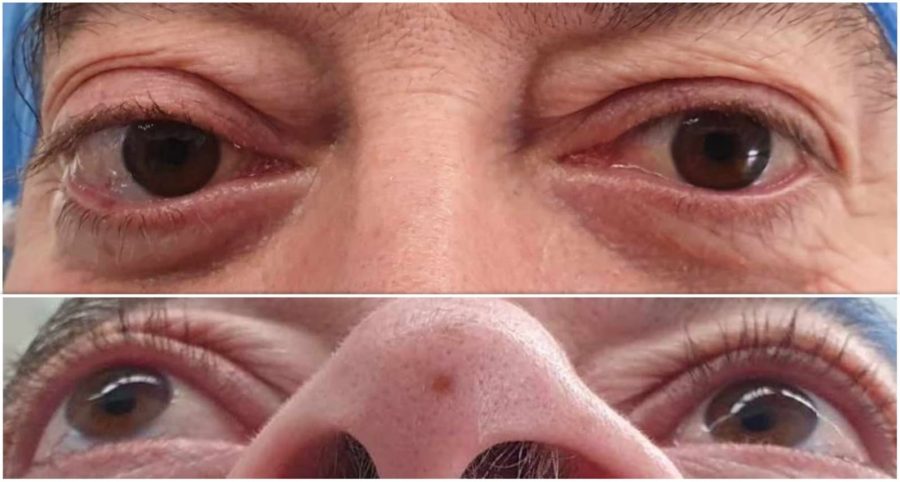
Figure 1: Photographs in orthostatic (top) and supine (bottom) positions illustrating the axial proptosis of the right eye, without relevant ocular deviation.
Case History
A 50-year-old Caucasian man was referred for diagnosis and treatment of a progressive, painless proptosis of his right eye (RE) over the course of 6 months. He had no complaints of pain, diplopia, or vision loss. There were no records of trauma. His general health and family history were unremarkable. On examination, uncorrected visual acuity (UCVA) was 10/10 in both eyes. The right lid crease was slightly larger than the left, with no lid retraction (Figure 1). There were neither signs of ocular surface inflammation, nor ulcers or scars. Fundoscopy and intraocular pressure were bilaterally normal. There was neither limitation of the ocular movements nor relative afferent pupillary defect. Exophthalmometry on the right was 25/20/3.5. Palpation of the globe offered some resistance to retropulsion, but no external mass was detectable. No pulse or bruit could be felt.
The magnetic resonance imaging (MRI) of the skull ordered by the referring doctor and brought by the patient displayed an intraconal orbital mass of heterogeneous hyperintensity, in close contact with the optic nerve, with no apparent disruption of the nerve sheath. Visual field testing rendered normal results.
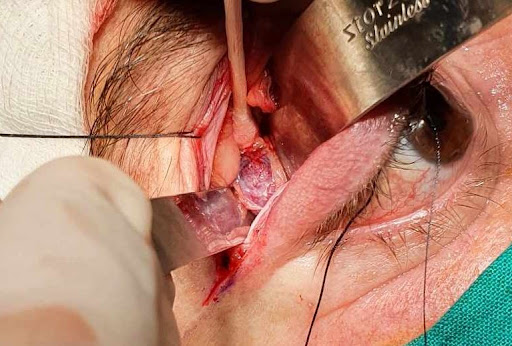
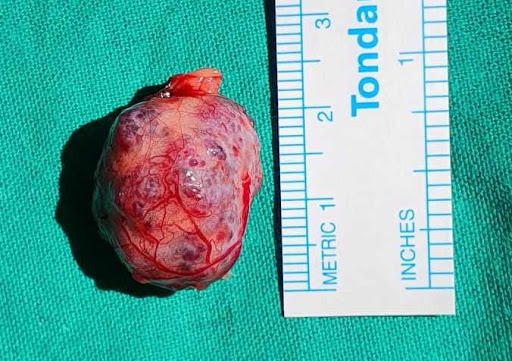
Surgical exploration for excisional biopsy was indicated. Through a transpalpebral superolateral orbitotomy (Figure 2), the mass was completely isolated and excised. Macroscopically, the tumor consisted of a lobulated, well-circumscribed, violaceous tissue growth, completely involved by a brilliant, smooth, translucid, highly vascularized capsule. The lesion measured 2.9 X 1.9 cm. The gross appearance was that of a vascular neoplasia, with no signs of necrosis or bleeding (Figure 3).
Figure 2: Surgical approach through a transpalpebral superolateral orbitotomy, allowing from direct visualization and total excision of the tumor.
Figure 3: Macroscopic aspect of the resected tumor, a roundish, lobulated mass of violaceous tissue of soft texture, measuring 2.9 cm in its longest axis.
The ectoscopic changes receded in the immediate postoperative period, with noticeable return of the ocular globe to its physiological position, with no functional or aesthetic sequelae.
Additional History
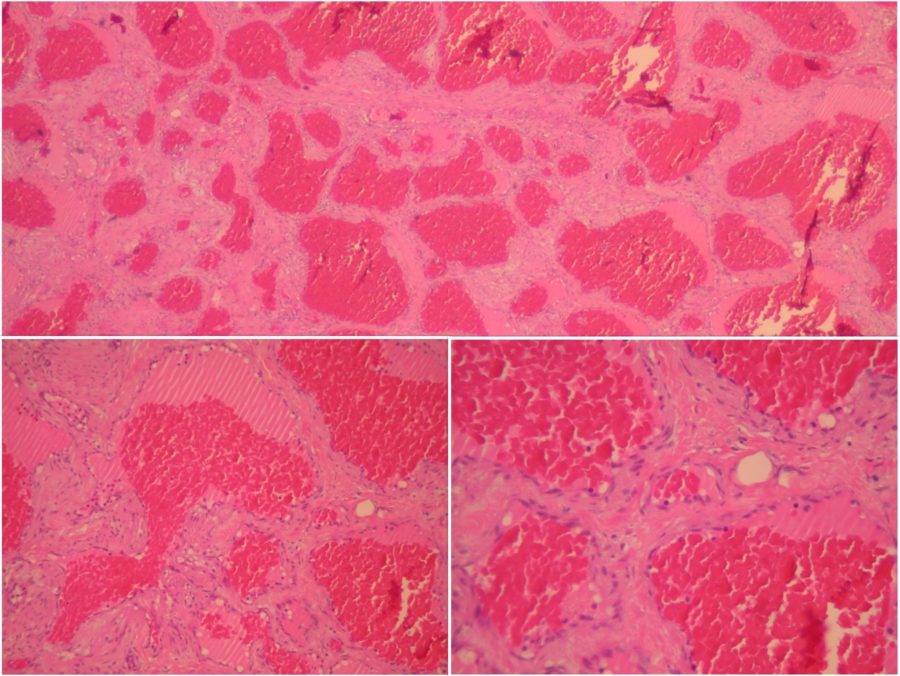
The excised lesion was sent to histopathological examination. Under microscopic examination, the neoplasm consisted of multiple vascular channels and abundant stroma. The vascular structures are filled with blood and variable degrees of thrombosis, confirming the main diagnostic hypothesis of orbital cavernous hemangioma (Figure 4).
Figure 4: Microscopic photographs (histopathological images) of the mass, with the characteristic features of cavernous hemangioma. Top: hematoxylin-eosin stain (H+E) at 25X magnification displaying thin-walled, abnormally dilated and hyperemic vessels in fibrotic stroma. Bottom left: the same area in higher magnification (100X). Bottom right: 200X magnification showing that the inner surface of the vessels is covered by flat endothelial cells without atypia.
Considering the benign nature of the tumor, the thorough surgical excision, and the restoration of the orbital normality, the patient was scheduled for regular follow-up, without further interventions.
Differential Diagnosis of Orbital Cavernous Hemangioma
- lymphoma
- lymphangioma
- hemangiopericytoma
- orbital inflammatory syndrome (orbital pseudotumor)
- Grave’s disease
- meningioma
- dermoid cyst
- fibrous histiocytoma
- neurofibroma
- orbital schwannoma
- orbital metastasis
Definitive diagnosis of any orbital neoplasm requires biopsy. It is nearly impossible to differentiate between these tumors based on clinical exam alone. An encapsulated tumor formed of an intricate net of slow-flow vascular structures is typical of orbital cavernous hemangiomas.
Discussion and Literature
Cavernous hemangioma (CH) is the most common primary orbital lesion of adults. Recently considered a type 3 low-flow arterial-venous malformation, orbital CH (OCH) occurs more often in women and typically presents in the fourth and fifth decades of life. It is a benign vascular growth characterized by a well-defined capsule and numerous large vascular channels. The most common sign of OCH is progressive axial proptosis from the preferential involvement of the intraconal orbital space. Optic nerve damage and other signs of orbital pathology may be present, with a variable degree of visual impairment.
There is no side predilection, but OCH are generally located at the temporal quadrant. Multifocal and bilateral occurrence has been described, sometimes associated with syndromes predisposing to hemangiomas. The progression rate is extremely variable. Symptoms may last for weeks to years and are nonspecific in the beginning. Pain is not common, but some patients may complain of frontal headache. Most of the cases present proptosis and orbital enlargement. Bruits or pulsations are rarely seen. Disc edema and choroidal folds are possible findings, but optic atrophy is rare. Diplopia and corneal compromise are uncommon. Intraocular tension is usually not affected.
B-Scan, computed tomography and magnetic resonance are the main imaging methods for the diagnosis of OCH. Surgical removal is recommended, at least in cases with constant growth tendency, marked bulbar protrusion or significant optic nerve compression. Imaging is paramount not only for differential diagnosis but also in planning the surgical approach. The majority of lesions are found within the muscle cone. In most cases, it is possible to dissect the cavernomas from the surrounding tissues since they are well encapsulated and occur as a single mass. Unlike the rarer intracranial cavernomas, OCHs have no tendency to bleed.
Micromorphologically, OCHs are benign vascular growths characterized by multiple large vascular channels covered by endothelial cells and abundant stroma. The vascular lumen is filled with blood and variable regions of intralesional thrombosis, reflecting vascular stasis/extremely slow flow. Unlike for subcutaneous and hepatic lesions, a well-defined and compact capsule is a typical feature of OCHs. Recent studies using immunohistochemistry, and the iteration for the classification of vascular lesions proposed by the International Society for the Study of Vascular Anomalies (ISSVA – 2018) suggest that these growths be classified as vascular malformations instead of neoplasia.
Possible complications of OCHs are acquired hyperopia of the affected eye, neurogenic or muscularly limited ductions (commonest in elevation), reduction of visual acuity, and visual field deficit due to direct compression of the optic nerve or its blood supply. Nevertheless, the prognosis of OCHs is usually good. The potential for malignization is virtually null. Smaller lesions without significant anatomical and functional distress can be safely kept under conservative follow-up. Recurrence has not been observed even for incomplete resection of masses located deep in the orbit.
Keep in mind
- In any unilateral, progressive proptosis, the possibility of an orbital tumor must be excluded.
- Orbital tumors may be benign or malignant. Prompt diagnosis allows for earlier resolution of symptoms and better prognosis.
- Large cavernous hemangiomas of the orbit may have aesthetic and/or functional implications, but surgical excision can be curative in most cases.
References
- Herter T, Bennefeld H & Brandt M (1988). Orbital cavernous hemangiomas. Neurosurgical review, 11(2), 143–147. https://doi.org/10.1007/BF01794679
- Calandriello L, Grimaldi G, Petrone G, Rigante M, Petroni S, Riso M & Savino G (2017). Cavernous venous malformation (cavernous hemangioma) of the orbit: Current concepts and a review of the literature. Survey of ophthalmology, 62(4), 393–403. https://doi.org/10.1016/j.survophthal.2017.01.004
- Sullivan TJ (2018). Vascular Anomalies of the Orbit–A Reappraisal. Asia-Pacific journal of ophthalmology (Philadelphia, Pa.), 7(5), 356–363. https://doi.org/10.22608/APO.2017151
- Tawfik HA & Dutton JJ (2022). Orbital Vascular Anomalies: A Nomenclatorial, Etiological, and Nosologic Conundrum. Ophthalmic plastic and reconstructive surgery, 38(2), 108–121. https://doi.org/10.1097/IOP.0000000000002029
- Ansari SA & Mafee MF (2005). Orbital cavernous hemangioma: role of imaging. Neuroimaging clinics of North America, 15(1), 137–158. https://doi.org/10.1016/j.nic.2005.02.009
- Zhang L, Li X, Tang F, Gan L & Wei X (2020). Diagnostic Imaging Methods and Comparative Analysis of Orbital Cavernous Hemangioma. Frontiers in oncology, 10, 577452. https://doi.org/10.3389/fonc.2020.577452
- Bonavolontà G, Strianese D, Grassi P, Comune C, Tranfa F, Uccello G & Iuliano A (2013). An analysis of 2,480 space-occupying lesions of the orbit from 1976 to 2011. Ophthalmic plastic and reconstructive surgery, 29(2), 79–86. https://doi.org/10.1097/IOP.0b013e31827a7622
- Mueller SK & Bleier BS (2022). Endoscopic surgery for intraconal orbital tumors. Endoskopische Chirurgie bei intrakonalen Orbitatumoren. HNO, 70(5), 345–351. https://doi.org/10.1007/s00106-022-01156-y
- Scheuerle AF, Steiner HH, Kolling G, Kunze S & Aschoff A (2004). Treatment and long-term outcome of patients with orbital cavernomas. American journal of ophthalmology, 138(2), 237–244. https://doi.org/10.1016/j.ajo.2004.03.011