Presented by: Paris Tranos MD, PhD, ICOphth

Edited by: Penelope Burle de Politis, MD

A 66-year-old woman was referred for investigation of yellowish-white fundus lesions in the right eye.
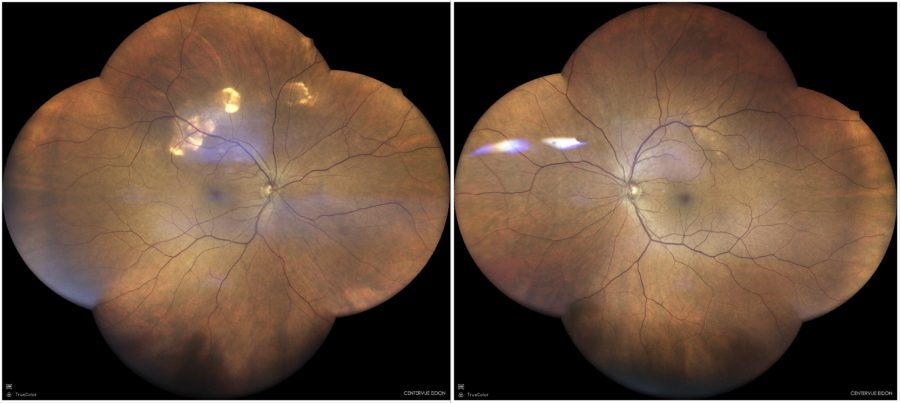
Figure 1: Color fundus photograph (EIDON true-color confocal scanner®) displaying temporal-superior yellowish-white lesions in both eyes, more evident in the right eye.
Case History
A 66-year-old Caucasian woman was referred for investigation of posterior pole lesions in the right eye (RE) noticed by chance during routine ophthalmological check-up. Her previous ocular history was unremarkable. Her past medical history included hypercholesterolaemia, rheumatoid arthritis, and hypothyroidism secondary to Hahimoto’s disease. There was no relevant family history. Upon examination, her corrected distance visual acuity (CDVA) was 8/10 in the RE and 10/10- in the LE. Refractometry was -1.50-1.25@110° in the RE and +2.25-1.00@105° in the LE, and intraocular pressure was within normal limits bilaterally. Anterior segment biomicroscopy revealed bilateral moderate nuclear cataract, denser in the RE. At fundoscopy, both eyes (BE) presented yellowish-white longitudinal deep lesions at the superior midperiphery and along the superotemporal vascular arcades, more pronounced in the RE (Figure 1). There were no cells nor sheets in the vitreous.
Spectral domain optical coherence tomography (SD-OCT) evidenced a characteristic rocky configuration of the lesions, with undulations of the retinal pigment epithelium (RPE), dilation of Haller’s layer in the choroid, and scleral thickening with focal compression of the choroid (Figure 2).
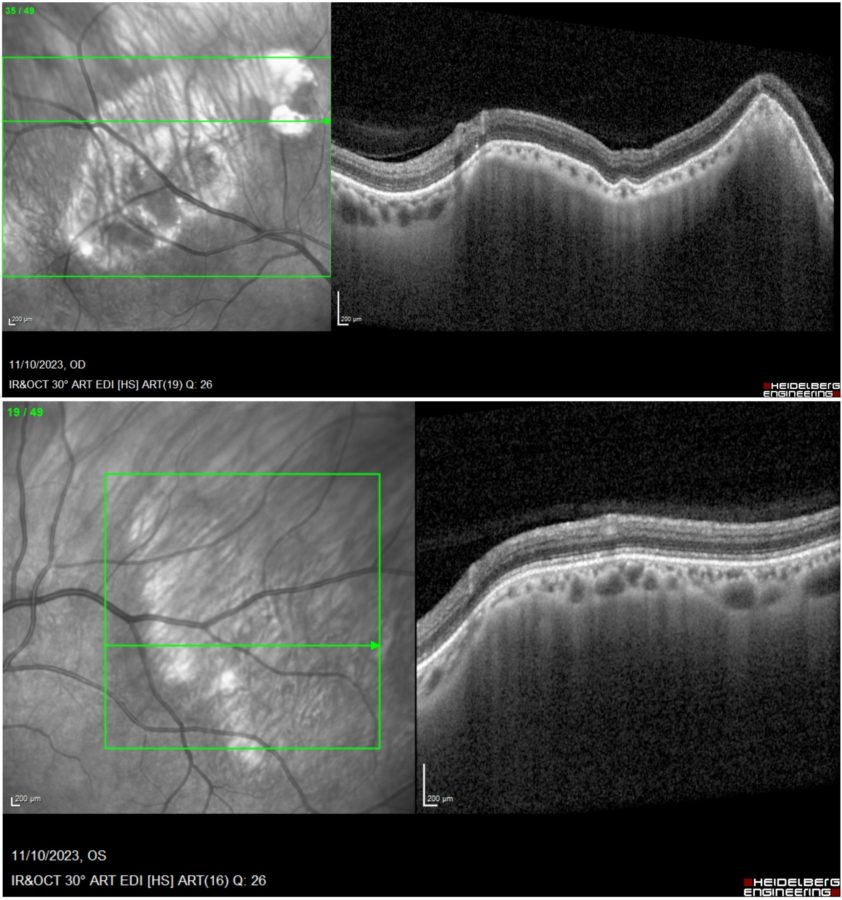
Figure 2: Infrared reflectance (IR) and enhanced depth imaging optical coherence tomography (EDI-OCT, Heidelberg Engineering®) demonstrating clusters of hyperdense lesions at the posterior pole (left top and bottom) and multiple elevations in both eyes, more numerous and exuberant in the right eye (top images).
Blue-light fundus autofluorescence (BAF) imaging displayed multiple hypoautofluorescent spots surrounded by hyperautofluorescent areas in the RE (Figure 3).
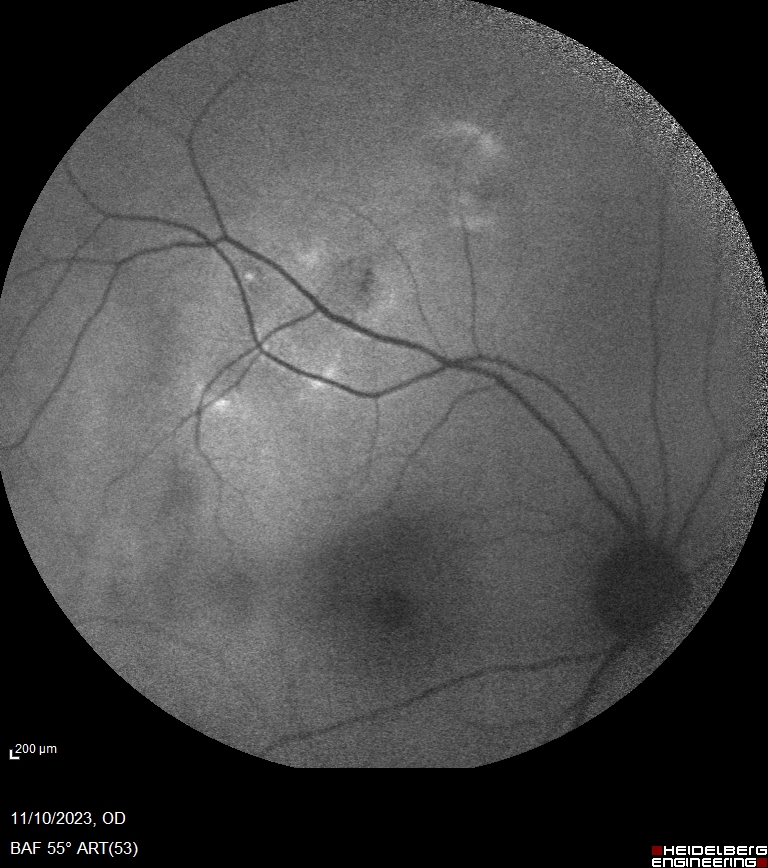
Figure 3: Blue-light fundus autofluorescence (BAF) photograph of the right eye (Heidelberg Engineering®) illustrating the hypoautofluorescent aspect of the lesions among hyperautofluorescent haloes.
Fundus Fluorescein Angiography (FFA) revealed hyperfluorescence of the lesions in the RE, more intense in the late phases (Figure 4).
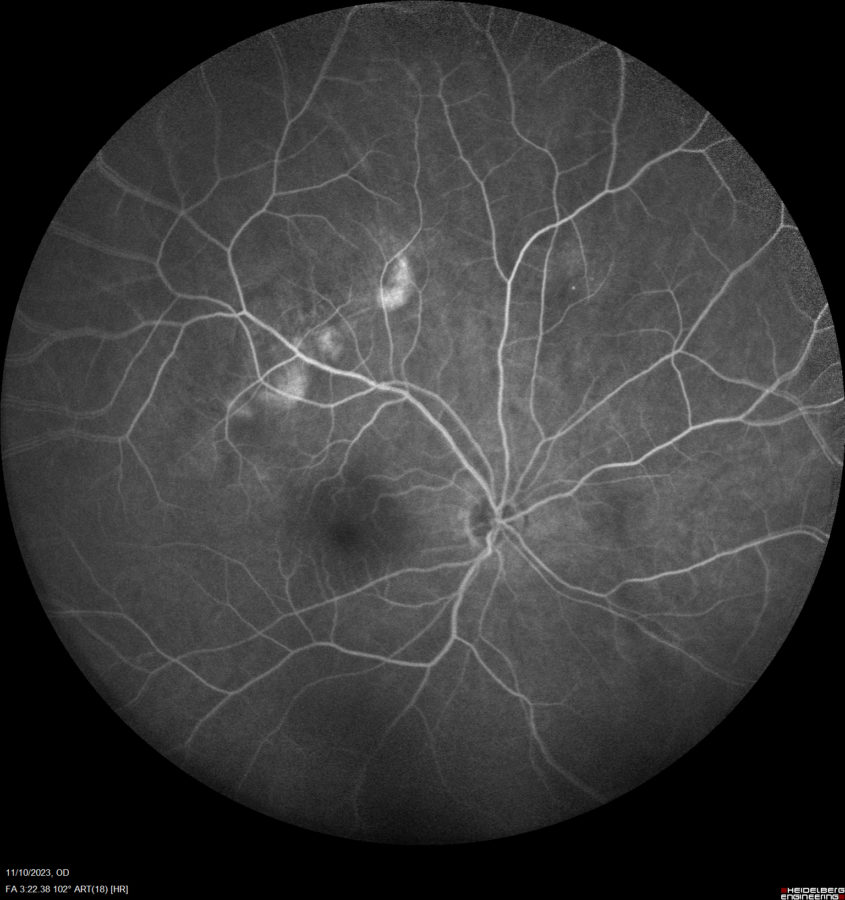
Figure 4: Fundus Fluorescein Angiography (FFA, Heidelberg Engineering®) exhibiting late hyperfluorescence of the lesions in the right eye.
Indocyanine green angiography (ICGA) showed sustained hypocyanescence of the lesions throughout the exam (Figure 5).
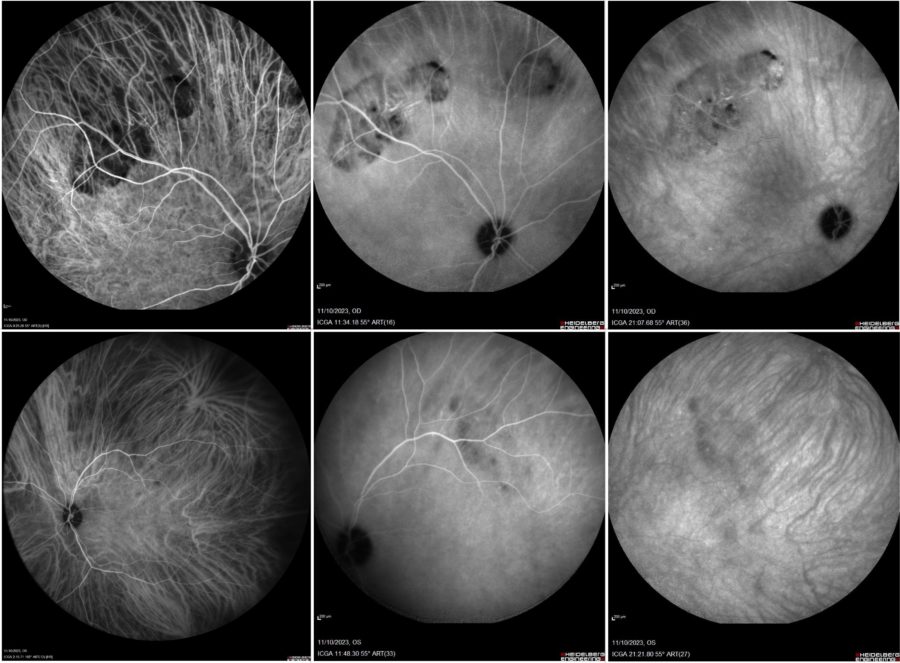
Figure 5: Indocyanine green angiography (ICGA, Heidelberg Engineering®) featuring hypocyanescence of the lesions from early to late phases (progressively left to right) in both eyes (top: right eye; bottom: left eye).
B-scan ultrasonography displayed hyperechogenic images with acoustic shadowing indicating calcified lesions in BE, more marked in the RE (Figure 6).
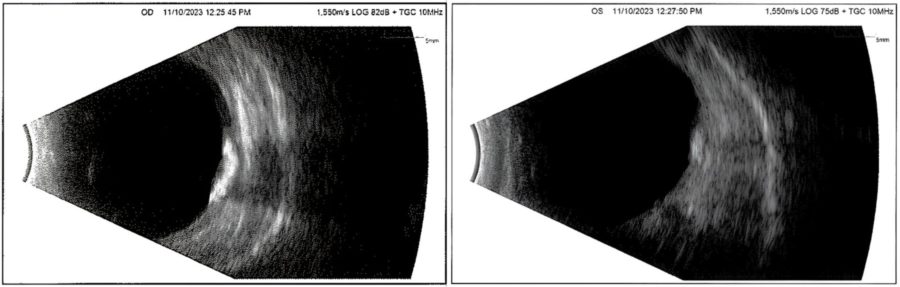
Figure 6: B-mode ultrasonography (respectively right and left eyes) showing characteristic hyper-echoic elevations with posterior shadowing at the posterior wall of both eyes, more pronounced in the right eye.
Additional History
Based on the clinical and multimodal image findings, the diagnosis of sclerochoroidal calcification was established.
Considering the benign nature of the lesions and lack of correlated visual compromise, the case was managed with conservative ophthalmological follow-up.
Further screening for underlying renal and/or parathyroid disease was advised.
Differential Diagnosis of Sclerochoroidal Calcification
- choroidal osteoma
- choroidal metastasis
- choroidal melanoma
- choroidal lymphoma
- amelanotic choroidal nevus
- choroidal granuloma
- choroidal hemangioma
- chorioretinitis
- regressed retinoblastoma
- retinal astrocytic hamartoma
The differential diagnosis between sclerochoroidal calcification and other fundus lesions is based on their distinct clinical presentation, fundus findings and multimodal imaging characteristics.
Discussion and Literature
Sclerochoroidal calcification (SCC) is a rare degenerative disorder characterized by calcium deposition in the sclera and/or choroid. SCC lesions characteristically appear as clear, uncircumscribed, yellowish-white, flat or minimally elevated placoid masses. Their typical localization is mid-peripheral, between the arcades and the equator, most frequently at the superotemporal quadrant, at the sites of insertions of the oblique extraocular muscles.
Idiopathic sclerochoroidal calcification may be easily overlooked or misdiagnosed. In the 1960s and 1970s, there were a few reports on the histopathology of ocular calcification in patients with parathyroid abnormalities. In 1982, fundus features of calcium deposition, labeled as “metastatic calcification,” were reported in a patient with hyperparathyroidism. The term idiopathic sclerochoroidal calcification was first mentioned in 1989, in a case report of bilateral multiple calcific choroidal foci with no hypercalcemia or any other ocular abnormalities.
SCC is usually seen in elderly patients. There seems to be no gender predilection, though a slight female predominance has been noted. Most reported patients are Caucasians. SCC is usually found as an asymptomatic incidental finding. Lesions may be unilateral or bilateral, unifocal or multifocal. There is frequently a halo of retinal pigment epithelium (RPE) atrophy surrounding the lesion. Visual acuity is usually not affected unless there is CNV, subretinal fluid, or macular involvement.
SCC manifests as an acoustically solid mass with posterior shadowing on B-mode USG and displays calcification on CT. On FA, SCC shows early venous onset of fluorescence and late hyperfluorescence. On OCT, the lesion has either a smooth or jutted appearance with scleral hyperreflectivity, and posterior shadowing consistent with calcium content. On OCT-angiography (OCT-A), SCC displays hyporeflectivity in the outer retina and choriocapillaris from posterior shadowing. Associated CNV and retinochoroidal anastomosis may occur and can be demonstrated by OCT-A.
Systemic work-up is important in patients with SCC. Although most cases are idiopathic, renal, endocrinologic, and skeletal pathologies leading to abnormal calcium and phosphorus metabolism should be excluded in all patients. While idiopathic cases tend to be seen in elderly patients, SCC associated with a systemic disorder is usually seen at younger ages. Both idiopathic SCC and cases associated with systemic disorders usually appear as bilateral and multiple lesions. Early detection of systemic diseases altering calcium metabolism are crucial to prevent or reduce ocular and systemic morbidity.
In general, SCC is a benign condition and remains stable. In a minority of cases, it may demonstrate slow progression and enlargement on long-term follow-up. CNV is the most common cause of vision loss associated with SCC and thought to be related to choroidal compression by the calcified masses, with reduction of oxygen supply to the choroid. CNV in SCC is associated with subretinal fluid, subretinal hemorrhages, exudates, and hemorrhagic RPE detachments.
There is no treatment available to ameliorate the degenerative effects of SCC on the sclera and choroid. However, since most cases are asymptomatic, SCC generally does not require treatment unless it is complicated. Secondary CNV can be observed or treated with laser photocoagulation, PDT, and/or intravitreal anti-VEGF injections if vision is affected. If CNV is far from the macula, close follow-up remains as the conservative option.
Keep in mind
- Sclerochoroidal calcification usually manifests as multiple yellow-whitish placoid lesions in the superotemporal midperiphery of asymptomatic older white individuals.
- Most cases of SCC are idiopathic, but all patients should be tested for systemic disorders involving abnormal calcium metabolism or renal function.
- Multimodal imaging is essential for accurate diagnosis of SCC and early detection of possible complications.
References
- Schachat AP, Robertson DM, Mieler WF, Schwartz D, Augsburger JJ, Schatz H & Gass JD (1992). Sclerochoroidal calcification. Archives of ophthalmology (Chicago, Ill. : 1960), 110(2), 196–199.
- Gündüz AK & Tetik D (2023). Diagnosis and Management Strategies in Sclerochoroidal Calcification: A Systematic Review. Clinical ophthalmology (Auckland, N.Z.), 17, 2665–2686. https://doi.org/10.2147/OPTH.S399058
- Shields CL, Hasanreisoglu M, Saktanasate J, Shields PW, Seibel I & Shields JA (2015). Sclerochoroidal calcification: clinical features, outcomes, and relationship with hypercalcemia and parathyroid adenoma in 179 eyes. Retina (Philadelphia, Pa.), 35(3), 547–554. https://doi.org/10.1097/IAE.0000000000000450
- Cooke CA, McAvoy C & Best R (2003). Idiopathic sclerochoroidal calcification. The British journal of ophthalmology, 87(2), 245–246. https://doi.org/10.1136/bjo.87.2.245
- Mitamura M, Kase S & Ishida S (2020). Multimodal imaging in sclerochoroidal calcification: a case report and literature review. BMC ophthalmology, 20(1), 248. https://doi.org/10.1186/s12886-020-01520-y
- Brahma VL, Shah SP, Chaudhry NA & Prenner JL (2017). Bilateral Idiopathic Sclerochoroidal Calcifications. The open ophthalmology journal, 11, 76–79. https://doi.org/10.2174/1874364101711010076
- Honavar SG, Shields CL, Demirci H & Shields JA (2001). Sclerochoroidal calcification: clinical manifestations and systemic associations. Archives of ophthalmology (Chicago, Ill. : 1960), 119(6), 833–840. https://doi.org/10.1001/archopht.119.6.833
- Lindstedt EW, van den Born LI, Veckeneer M & Baarsma GS (2007). Sclerochoroidal calcification: idiopathic or associated with systemic disease?. Retinal cases & brief reports, 1(3), 141–144. https://doi.org/10.1097/01.ICB.0000279643.99324.93
- Sugarman JA, Douglass AM, Say EA & Shields CL (2017). Stones, bones, groans, thrones, and psychiatric overtones: Systemic associations of sclerochoroidal calcification. Oman journal of ophthalmology, 10(1), 47–49. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5338055/
- Abouzaid M, Al-Sharefi A, Artham S, Masri I, Kotagiri A & Joshi A (2019). A rare ophthalmic condition associated with primary hyperparathyroidism (PHPT): sclerochoroidal calcification (SC). Endocrinology, diabetes & metabolism case reports, 2019(1), 19-0003. https://doi.org/10.1530/EDM-19-0003


